Has the time come for a revival of the “cell state” perspective? Jonathan Birch looks at the application of social evolution theory to the origins of multicellular life.
The “cell state” metaphor
In the 19th century, biologists came to appreciate for the first time the fundamentality of the cell to all life on Earth. One of the early pioneers of cell biology, Rudolf Virchow, realized that the discovery of the cell brought with it a new way of seeing the organism. He described the organism as a “cell state”, or Zellenstaat, a “society of cells, a tiny, well-ordered state, with all of the accessories – high officials and underlings, servants and masters, the great and the small”.1
Virchow’s vision was widely supported in late 19th Century German biology. Ernst Haeckel, an influential early advocate of Darwinism, advocated the “cell state” metaphor throughout his career, reflecting towards the end of it that:
“The conception of cells as ‘elementary organisms’ led to the further opinion that our own human organism, just like all higher animals and plants, is actually a ‘cell state’, composed of millions of microscopic citizens, the individual cells, which work more or less independently, and co-operate for the common purpose of the entire state.”2
In Britain, meanwhile, the philosopher Herbert Spencer coined the notion of the “social organism”, a complex, two-way analogy between the cells of the body and the members of a society:
“Hence we are warranted in considering the body as a commonwealth of monads, each of which has independent powers of life, growth, and reproduction; each of which unites with a number of others to perform some function needful for supporting itself and all the rest; and each of which absorbs its share of nutriment from the blood. And when thus regarded, the analogy between an individual being and a human society, in which each man, whilst helping to subserve some public want, absorbs a portion of the circulating stock of commodities brought to his door, is palpable enough.”3
In the 20th Century, the “cell state” metaphor fell out of favour in biology. The rise of biochemistry and, later, molecular biology was accompanied by the emergence of a new metaphor: the notion of a cell as a factory, subordinate to the organism as a whole and dedicated to serving its biochemical needs.4 Has the time now come for a revival of the “cell state” perspective? Three trends in recent social evolution research suggest that it has.
Firstly, there is our growing awareness of the scale of cooperation among microorganisms (see Parts 1 and 3). Some social behaviours in microbes, such as the formation of a grex in social amoebas, result in phenomena that clearly resemble simple multicellular organisms. More controversially, some authors, such as Marc Ereshefsky and Makmiller Pedroso, have suggested that even biofilms should be regarded as multicellular individuals in their own right.
Secondly, in insect biology, there has been a revival of interest in the “superorganism” concept: the suggestion that we should think of an insect colony as a single, higher-level organism. While this idea has always been controversial, even to consider it is to recognize the possibility that a high degree of social complexity can in principle give rise to a new, organism-like entity.
Thirdly, a research program called “major transitions theory” has led to a radical re-evaluation of the place of cooperation in the history of life. Building on foundations laid by, among others, Leo Buss, John Tyler Bonner, and John Maynard Smith and Eörs Szathmáry, major transitions researchers portray the history of life as a series of “evolutionary transitions in individuality” in which integrated, higher-level individuals have evolved, through social evolution, from groups of smaller entities.
When we look at evolution in this new light, we start to see social phenomena where we saw none before. We see cooperation among cells within multicellular organisms, among organelles within cells, even among genes within a chromosome. The result has been a dramatic increase in the explanatory ambitions of social evolution research – and the return of “cell state” thinking.
Importantly, for social evolution theorists working on the origins of multicellular life, the “cell state” is more than a metaphor: it’s the foundation for a research program. In other words, taking a “social perspective” on the multicellular organism does not simply mean describing the activities of cells in social terms. It means drawing on the concepts and methods of social evolution theory to explain the evolutionary origins of multicellular individuals.
Hamilton’s hypothesis
For one of the pioneers of major transition theory, Leo Buss, the evolution of multicellular life presented the following puzzle: given that natural selection favours cells that promote their own reproduction, how did early multicellular organisms avoid being destabilized by conflict among cell lineages?5
His answer was that such organisms have evolved – through group selection – mechanisms for controlling internal conflict, such as “germline segregation”, whereby the capacity to generate a new organism is limited to a very small number of cell lineages (the germline) that are set aside early in development. This, however, led to a “chicken and egg” puzzle: how could group selection be powerful enough, relative to individual selection, to assemble such mechanisms prior to the existence of such mechanisms?
However, when we view the organism through the lens of inclusive fitness, this puzzle dissolves. Multicellular organisms are clonal groups, and there is no reason for the cell lineages in a clonal group to be in evolutionary conflict. A clonal group is one in which the conditions are highly favourable to the evolution of altruism. As Hamilton himself observed:
“Our theory predicts for clones a complete absence of any form of competition which is not to the overall advantage and also the highest degree of mutual altruism. This is borne out well enough by the behavior of clones which make up the bodies of multicellular organisms.” 6
In other words, there is no big puzzle about the stability of cooperation among the cell lineages of the first multicellular organisms. These organisms were clonal groups, and they cooperated because they were genetically near-identical. No further mechanisms were needed to suppress competition, so no “chicken and egg” problem arises. Let’s call this “Hamilton’s hypothesis”.
Queller has taken this hypothesis further, arguing that relatedness can even explain the origins of a “germ-soma” division of labour. He suggests that “the germ line might have originated as a consequence of other cell lineages’ altruistically removing themselves from the reproductive line, to perform some somatic benefit to the organism”.
This turns the explanatory relationship posited by Buss on its head. Far from being a group-level adaptation for suppressing conflict among cells, the germline, on Queller’s account, arose as a by-product of altruism – altruism that evolved because the inclusive fitness interests of the cells were aligned.
Complications
Hamilton’s hypothesis is complicated by the fact that few organisms are fully clonal. Clonality is a feature of organisms that develop from a single cell, but not all development is like this. For example, many plants can reproduce vegetatively, with the new individual developing from a multicellular offshoot.
In plants under cultivation, many generations of vegetative reproduction lead to the accumulation of mutations in different cell layers, resulting in individuals containing internal genetic diversity. In the wild, though, plants frequently reproduce sexually, pushing their lineage through a single-cell bottleneck and restoring high relatedness. Provided this happens often enough, high relatedness and low internal conflict will be maintained.
Even among organisms that pass through a single-cell bottleneck every generation, such as multicellular animals, clonality is not perfect. Within-organism genetic diversity can arise through “chimerism”, where cell lineages produced from a different sperm and egg mix in the early stages of development. We see this, for example, in marmosets.
Genetic diversity also arises through mutation. Sometimes, these mutation events lead to cancer, and the survival of the population depends on these cancerous “cheater” lineages being unable to spread between organisms. We see a grim illustration of this point in the Tasmanian devil, now threatened with extinction due to an epidemic of facial tumours. Given this, we should not be surprised to find that an ability to discriminate “self” from “non-self”, and to attack intruding cell lineages, is present even in sponges, often regarded as among the simplest multicellular animals.
Yet, for all these caveats, it remains plausible that, for any organism spawned from a single cell and of a size within certain bounds, relatedness is high enough to stabilize cooperation among the cells. It’s also plausible that organisms which can reproduce vegetatively in the wild pass through a single-cell bottleneck often enough to generate enough relatedness to stabilize cooperation among their cells. So Hamilton’s hypothesis has, I think, stood the test of time. The ultimate explanation for the stability of cooperation within multicellular organisms, at least up to a certain size, is genetic relatedness.
Open questions
So what still needs explaining about the origins of multicellular life, from the point of view of social evolution theory? Many of the most intriguing questions concern the process by means of which a social group of cells, once formed, is transformed over evolutionary time into a multicellular organism with a clear division of labour among distinct cell types. Andrew Bourke has called this process “social group transformation”.7
One open question here concerns the relationship between the size of a multicellular organism and its complexity. In the natural world, we find a clear positive correlation between the total number of cells an organism contains and the number of distinct, specialized cell types it contains. What explains this relationship?
Another concerns the limits on the number of cell types. There may have been as many as 25 separate instances of the evolution of multicellular life, but in only three of these cases – the plants, animals, and fungi – has the lineage in question proceeded to evolve large numbers of distinct cell types. It seems that evolving high numbers of specialized cell types remains improbable even once a lineage has attained evolved multicellularity. Indeed, it suggests that special further conditions are required. What are these conditions?
For now, I will simply leave these questions open. Some possible answers are developed in Chapter 7 of The Philosophy of Social Evolution.
← Part 3Part 5 →
Jonathan Birch is Assistant Professor in the Department of Philosophy, Logic and Scientific Method, specializing in the philosophy of the biological sciences. Most of his work concerns the evolution of social behaviour. He is also interested in the evolution of morality, animal sentience, and the relation between sentience and welfare.
Notes
[1] R. Virchow, 1859, ‘Atoms and Individuals’, in L. J. Rather, ed., Disease, Life, and Man: Selected Essays by Rudolf Virchow, Stanford University Press, p. 124.
[2] Quoted in A. Reynolds, 2008, ‘Ernst Haeckel and the Theory of the Cell State: Remarks on the History of a Bio-Political Metaphor’, History of Science, 6, p. 128
[3] H. Spencer, 1851, Social Statics, 1st ed., John Chapman, p. 451.
[4] Reynolds, op. cit.
[5] L. Buss, 1987, The Evolution of Individuality, Princeton University Press.
[6] W. D. Hamilton, 1964, ‘The Genetical Evolution of Social Behaviour’ I and II, Journal of Theoretical Biology, 7, p. 25.
[7] A. F. G. Bourke, 2011, Principles of Social Evolution, Oxford University Press.
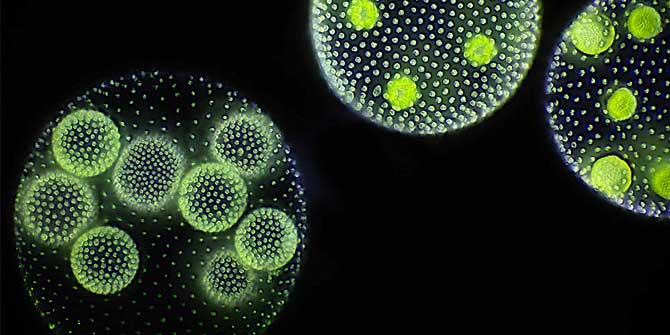
Featured image: Multicellular green algae of the Volvox genus (Micrograph by Frank Fox at www.mikro-foto.de, CC BY-SA 3.0 Germany licensed)