In the first installment of this five-part series, Jonathan Birch looks at biological altruism, a key concept from his new book The Philosophy of Social Evolution.
What is biological altruism? We tend to think of altruism as action motivated by concern for others. Biologists, however, use the term in a different way. In biology, altruism is, roughly speaking, behaviour that has two distinctive consequences for reproduction: it reduces the chances of reproduction of the organism that performs the behaviour, and it increases the chances of reproduction of another organism.1 The easiest way to grasp this idea is through a few examples.
Insects
Ants, wasps and bees are everyone’s favourite biological altruists. In species known as “eusocial”, there is a division of labour between the workers and the queen: the workers sacrifice their own chances of reproduction to raise the queen’s larvae. Some ants become living “honeypots”, hanging upside down from the roof of the nest, storing food for the rest of the colony. An especially poignant example of self-sacrifice is found in a species of Brazilian ant, Forelius pusillus. At the end of each day, some of the workers will seal the entrance to the nest from the outside, protecting it from nocturnal predators—but dooming themselves to die in the cold night-time temperatures.
One might object: is this really biological altruism, if the workers are sterile? I said biological altruism reduces an organism’s chances of reproducing—but doesn’t a sterile worker ant have zero chance of reproduction no matter what it does? In fact, only in a few species of ant are the workers fully sterile. In most species, workers can produce eggs which (though unfertilized) can develop into males—but the vast majority never activate their ovaries. In those species in which the workers are fully sterile, they are still biological altruists, but we should think about their altruism in a different way. We should say that these ants’ entire developmental process, rather than any specific behaviour, is altruistic. The whole process of developing into a sterile worker confers benefits on others at a cost to self.
Microbes
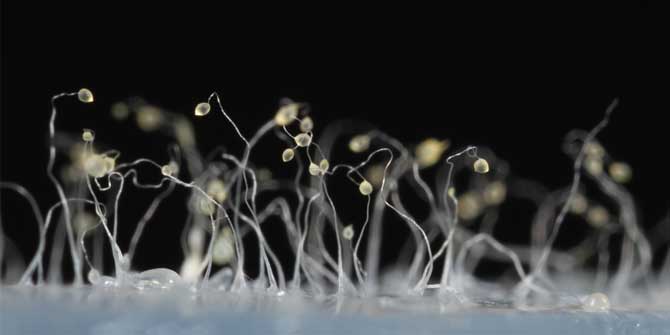
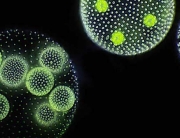
Some of the most spectacular acts of self-sacrifice in nature are too small for the naked eye to see. Consider the social amoeba Dictyostelium discoideum. For much of their life cycle, these amoebae live as single-celled organisms. But when food gets scarce, the amoebas start signalling to their neighbours. If they find out that other amoebas are present in sufficient numbers, the starving amoebas aggregate to form a mobile slug called a “pseudoplasmodium” or “grex”. The grex moves as one, further and faster than any individual amoeba could, in the direction of heat and light. On reaching a favourable location, the slug stops and begins to transform into a fruiting body.
Around a fifth of the amoebas sacrifice their lives in this process, forming a hardy, cellulose stalk of dead cells. The remaining four fifths cluster at the tip of the stalk, where they generate and release spores. The spores are dispersed through the environment, reducing the chances that the amoebas they ultimately produce will encounter the same harsh conditions their parents endured.
It turns out that cooperation, albeit usually less dramatic than this, is pervasive in microbial populations. We now realize that what looked like a blob on a Petri dish is in reality a dynamic social network: a community in which vast numbers of microorganisms, often including members of several different species, interact with each other in complex ways. Perhaps the most frequent type of cooperative interaction involves the production and consumption of so-called “public goods”. These goods include enzymes, surfactants, antibiotics, adhesive polymers, ammonia, and other useful molecules. What they have in common is that, when emitted by a producer into the external environment, they confer a benefit on at least some other nearby microorganisms.
Humans
Humans, in their best moments, are capable of matching the heroism of ants and amoebas. Tales of ant-like self-sacrifice are the stuff of legend in many cultures. When the pioneering evolutionary theorist John Maynard Smith wanted a historical example to add colour to his model of the evolution of signalling, he reached for the tale of Philip Sidney, an Elizabethan aristocrat mortally wounded at the Battle of Zuphen. The legend goes that, as he lay dying, he handed his water to another wounded soldier, remarking “thy necessity is yet greater than mine”. He did not turn his abdomen into a living water tank and spend his life hanging from the ceiling, drip-feeding others, but he still deserves some credit.
For many Britons, the iconic example of self-sacrifice is that of Captain Oates, a member of the ill-fated British Antarctica Expedition of 1910–13. Suffering from gangrene and frostbite, and seeing that he was impairing the survival chances of the rest of the expedition, he left the tent and walked into a blizzard, remarking “I am just going outside and may be some time”. Ants of the species Temnothorax unifasciatus do something similar. When infected by a fungal pathogen, they leave the nest to die alone, minimising the risk of transmission of the disease to nestmates.
Prosocial behaviour
Such acts are the exception rather than the rule in human social life. But while extreme self-sacrifice may be unusual, an everyday, low-key form of “prosocial” behaviour is the glue that holds human societies together. Just as an ant colony relies on countless workers unspectacularly bringing food to the nest, and a bacterial biofilm relies on the sharing of molecular “public goods”, human societies are held together by the fact that we are not all free riders. We help each other. We do public-spirited things like paying taxes, doing voluntary work, and voting in elections.
In modern societies, just as in some ant colonies, some of these behaviours are coerced: we’d be punished if we didn’t do them. Moreover, we have reputations to maintain, and sometimes our reputation would suffer if we didn’t do these things. But this is not true in all cases, and it is not a sufficient explanation for human social behaviour. There is a general human tendency towards prosociality that can still be seen in most people even when coercion and reputational benefit is stripped away. We see it in such real-world phenomena as anonymous charity donations and donations of blood and tissue, as well as in simple economic experiments.
Extreme heroism aside, is public-spirited behaviour biologically altruistic? Does it really detract from your chances of reproducing? Sometimes it does: organ donations by living donors are an obvious example. It’s less obvious that taking a financial hit, as in the case of a charity donation, is biologically altruistic. In some cases, it may even increase your chances of reproducing by improving your attractiveness to potential sexual partners. Whether or not a behaviour is biologically altruistic depends on its long-term effect on an organism’s chances of reproduction over the course of its entire lifetime, and that can be hard to assess. This is why the term “prosocial” is helpful. Many everyday human behaviours are clearly “prosocial” in a broad sense, whether or not they are biologically altruistic.
The search for common principles
Are there common principles powerful enough to help explain biological altruism and prosocial behaviour wherever we find it in the natural world—in amoebas, ants, humans and beyond? My book The Philosophy of Social Evolution explores this question. My answer, in short, is that relatedness is the crucial unifying concept, and W. D. Hamilton’s theory of inclusive fitness tells us how to use relatedness to make sense of social behaviour. Inclusive fitness is a familiar idea for many biologists – less so for philosophers – but the application of this idea to microbial evolution and cultural evolution introduces new challenges.
In this series of blog posts, I’ll introduce some of the main ideas from the book.2 In Part 2, I’ll present the basic idea of inclusive fitness and explain why it matters. In Part 3, I’ll consider some of the complications that arise when applying inclusive fitness to evolution in microbes. In Part 4, I’ll discuss the idea that a multi-celled organism is itself a form of social group – a social group of cells, or “cell state” – and ask whether social evolution theory can help make sense of why multicellular organisms exist. In Part 5, I’ll turn to humans, and argue that inclusive fitness has a useful role to play in theories of cultural evolution.
Part 2 →
Jonathan Birch is Assistant Professor in the Department of Philosophy, Logic and Scientific Method, specializing in the philosophy of the biological sciences. Most of his work concerns the evolution of social behaviour. He is also interested in the evolution of morality, animal sentience, and the relation between sentience and welfare.
Notes
[1] These blog posts will introduce the ideas in a non-technical way. Readers looking for a more detailed introduction to some of the ideas in the book may want to take a look at Jonathan Birch, 2017, ‘The Inclusive Fitness Controversy: Finding a Way Forward’, Royal Society Open Science 4:170335.
[2] These blog posts will introduce the ideas in a non-technical way. Readers looking for a more detailed introduction to some of the ideas in the book may want to take a look at Jonathan Birch, 2017, ‘The Inclusive Fitness Controversy: Finding a Way Forward’, Royal Society Open Science 4:170335.
Featured image: Fruiting bodies of the social amoeba Dictyostelium discoideum (Photograph by Usman Bashir, CC-BY-SA 4.0 licensed)